Partners involved in Work Package (WP) 4 are focusing on the research and development (R&D) and optimisation of renewable energy sources (RES) for electrification. This comprehensive task begins with the technical assessment of the RES requirements for each integrated case study flowsheet. In parallel, other research teams are conducting R&D for electricity generation, storage, and supply from RES. Additionally, several partners are overseeing the integration of renewable energy in the selected flowsheets of the electrochemical toolbox, as well as the assessment of RES-based system scenarios for future large-scale deployment. The main concept of the process is illustrated in the following image.
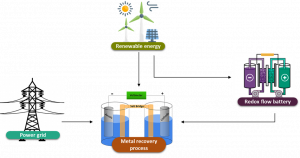
End goal of the WP4. Metal recovery process coupled with RES and a redox flow battery
Technical assessment of RES requirements of integrated case study flowsheets
The team at UCLM, responsible for the assessment of the RES requirements of the integrated case study flowsheets, is working to evaluate the best solutions to power the different flowsheets developed in WP 3 using RES. They are formulating models to understand the mass and energy balance, as well as the dynamics of the electrochemical cells and energy systems. This involves developing simulators to test and validate the models, followed by parameter estimations and process optimisation.
With the experimental data from TECNALIA’s electro-leaching technologies, researchers have reported encouraging metal recovery rates: 80% for vanadium (V), 77% for molybdenum (Mo), and 22% for tungsten (W). In the case of VITO’s gas diffusion electro-crystallization technology (GDEx), researchers reported metal recovery rates higher than 95 % for some Platinum Group Metals (PGMs) such as platinum (Pt), palladium (Pd) and rhodium (Rh). The energy consumption of both processes has been measured and optimised to ensure efficient use of RES reaching values of 72 kWh·kg-1 and 15 kWh·kg-1, for the electro-leaching and GDEx technologies, respectively. This assessment will be replicated for the other technologies explored in the FIREFLY project.
R&D for electricity generation, storage, and supply from RES
This task focuses on the feasibility of integrating RES with FIREFLY technology flowsheets, including the assessment of vanadium redox flow batteries for renewable energy storage. UCLM team in collaboration with LPRC have been collecting data about the wind and solar profiles in different localizations (La Palma and Ciudad Real, Spain) to optimise the power energy generation through RES. On the other hand, researchers have been conducting experiments under discharge conditions and carried out power energy estimation of solar and wind sources. They evaluated the system’s efficiency using various parameters for voltage and energy efficiency. Researchers will continue exploring the charge and discharge processes of batteries based on wind profiles.
R&D for RES integration in selected flowsheets of the electrochemical toolbox
This comprehensive task aims primarily to analyse various alternatives for the electrical design as well as their flexibility. The research entails multi-energy system analysis, flexibility potential analysis, and design options for electric power supply systems. ENGIE-LAB, responsible for this particular task, distributed an initial questionnaire to collect electrical information about selected electro-chemical processes. Given the early stage of the technologies development, more data needs to be input to draw more relevant conclusions.
Assessment of RES-based system scenarios for future large-scale deployment
Within this task, researchers are analysing the cost and emission savings impact of RES-driven processes developed in FIREFLY, as well as their applications in producing chemicals and processing biomass. The team at EnergyVille mapped the global demand and supply landscape of PGMs and highlighted the importance of recycling and integration of sustainable practices.
Additionally, researchers at EnergyVille conducted a techno-economic assessment of the gas-diffusion electrocrystallisation (GDEx) process, technology engaged to recycle PGMs from automotive catalysts. This assessment showed lower recovery costs (around 18,500 €·kg-1) compared to traditional recovery processes (20,000 – 50,000 €·kg-1), mainly due to the electrification of this process.
FIREFLY project was launched and it has been operating at times when Europe needs to leverage insights from R&D towards more sustainable chemical operations.
© visual UCLM
